ULI262
Developing a novel therapeutic
for people with FAP
The problem
No drug to prevent colorectal cancer
Curileum discovered and is developing a novel, oral small molecule drug that addresses the root cause of colorectal cancer: mutations leading to abnormal cell production forming polyps and progressing to cancer.
Colorectal cancer is one of the most common and deadly cancers worldwide. Current strategies for colorectal cancer prevention rely on periodic colonoscopies and surgical removal of polyps, which are costly, invasive and burdensome for patients. Despite screening programmes, many high-risk individuals still develop cancer.

Contact us to discuss potential investment opportunities
The problem
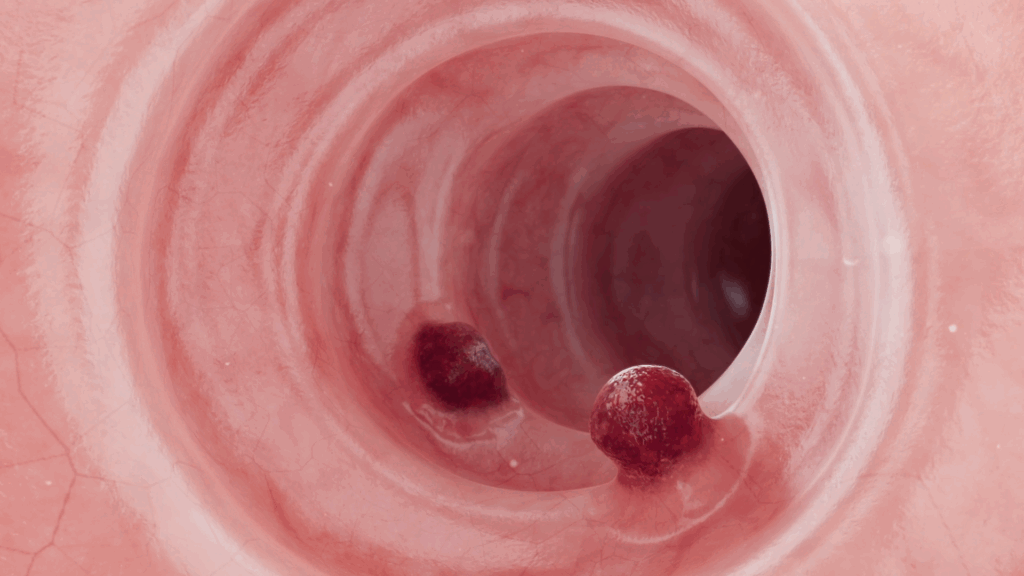
It starts with a polyp, no effective medical treatment
Our solution
ULI262- a first-in-class small molecule drug
Our first-in-class pan-PPAR agonist, ULI262, addresses these large, unmet needs in colorectal cancer prevention, targeting FAP patients, high-risk individuals identified in screening programmes, and post-cancer treatment prone to relapse.
By re-directing precancerous cells toward normal tissue, ULI262 may prevent progression to cancer – offering the first scalable, non-invasive pharmacological solution for colorectal cancer prevention.
Curileum is currently preparing for first-in-human clinical studies to treat patients with FAP. We expect the FDA will grant Orphan Drug Designation for the treatment of FAP, helping to expedite the clinical trial process and accelerate the timeline to treating patients in urgent need.